 The number and arrangement of valence electrons determine the chemical properties of an element. How Valence Electrons Determine Chemical Properties Understanding the behavior of valence electrons is essential in explaining the chemical properties and reactivity of elements and compounds. 
These are the electrons located in the outermost energy level or shell of an atom. 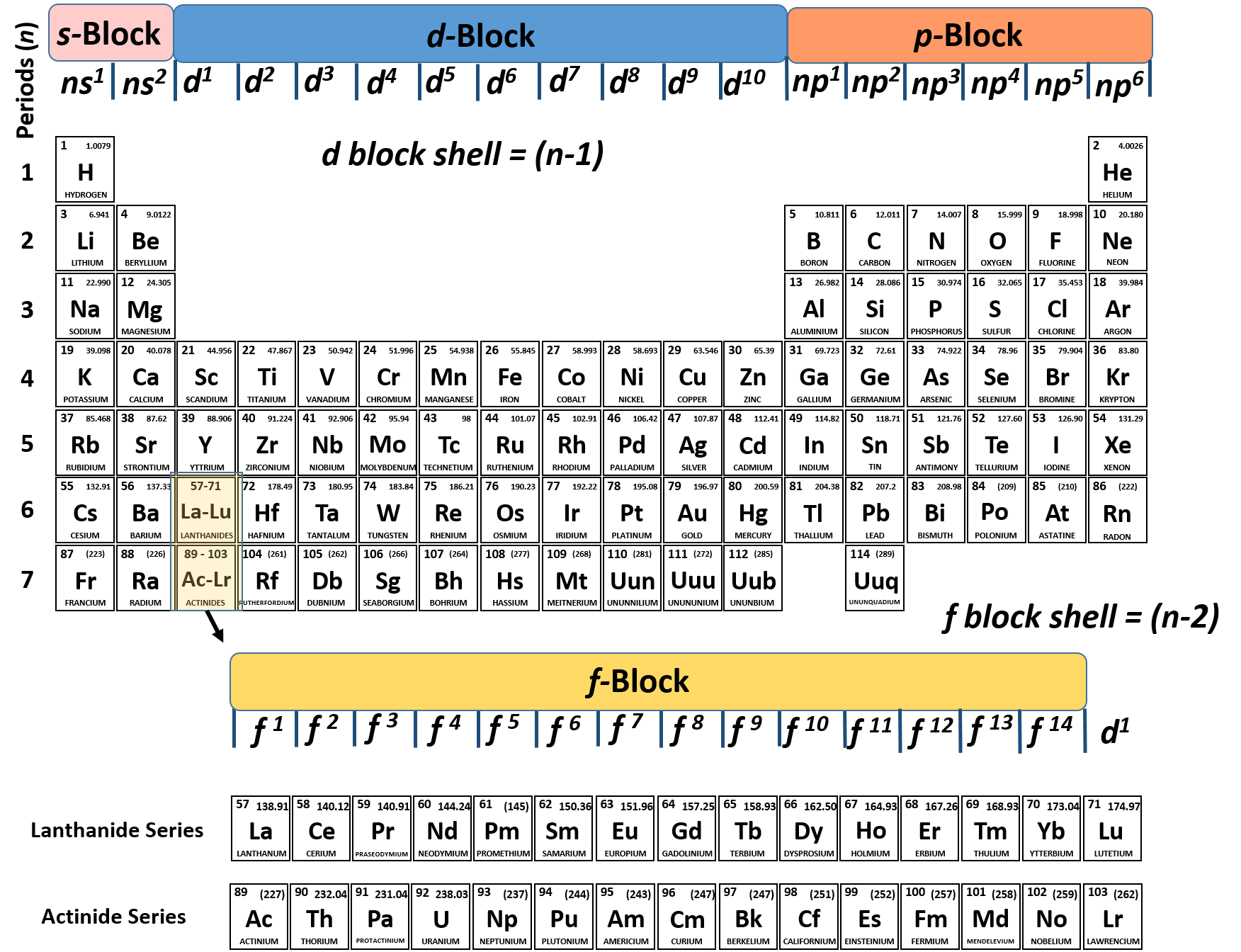
The Role of Valence Electrons in Chemical ReactionsĪdvertisements Valence electrons play a crucial role in chemical reactions. By considering the electron configuration and the position of an element in the periodic table, we can determine the number and location of valence electrons in an atom. They determine the type of chemical bond that can be formed and influence the reactivity of elements. In conclusion, valence electrons are crucial for understanding chemical bonding and the behavior of atoms. This means that carbon has 2 electrons in the first shell and 4 valence electrons in the second shell. For example, in the case of carbon ( atomic number 6), the electron configuration is 2,4. The valence electrons are found in the highest-numbered shell or energy level. For example, the first shell can hold a maximum of 2 electrons, the second shell can hold a maximum of 8 electrons, and so on. The maximum number of electrons that each shell can hold is determined by the formula 2n^2, where n is the shell number. The valence electrons are located in the outermost electron shell or energy level of an atom.Īdvertisements The electron shell closest to the nucleus is called the first shell, followed by the second shell, and so on. To find the valence electrons in an atom, you need to consider the electron configuration and the position of the element in the periodic table. Where to Find Valence Electrons in an Atom For example, all elements in Group 1 (alkali metals) have one valence electron, while elements in Group 18 ( noble gases) have a full complement of valence electrons. Elements in the same group or column of the periodic table have similar valence electron configurations. The periodic table provides a useful tool for determining the number of valence electrons in an atom. The electron configuration of an atom, which includes the arrangement of valence electrons, influences its physical and chemical properties. They determine the stability and chemical behavior of an atom. Valence electrons are essential for understanding the atomic structure and chemical reactivity of elements. Whether it’s a covalent bond, where electrons are shared between atoms, or an ionic bond, where electrons are transferred from one atom to another, the valence electrons are at the heart of these interactions. The valence electrons are crucial in determining the type of chemical bond that can be formed between atoms. The number of valence electrons determines how an atom will interact with other atoms to form chemical bonds. They are responsible for the chemical properties and behavior of an element. Valence electrons are the electrons located in the outermost energy level of an atom. In this article, we will explore the definition of valence electrons, understand their importance, and learn where to find them in an atom. 
These electrons are found in the outermost energy level or shell of an atom and are involved in the formation of chemical compounds. Valence electrons play a crucial role in chemical bonding and the reactivity of atoms. Understanding Valence Electrons Image by Samsiq – Wikimedia Commons, Wikimedia Commons, Licensed under CC BY-SA 4.0. 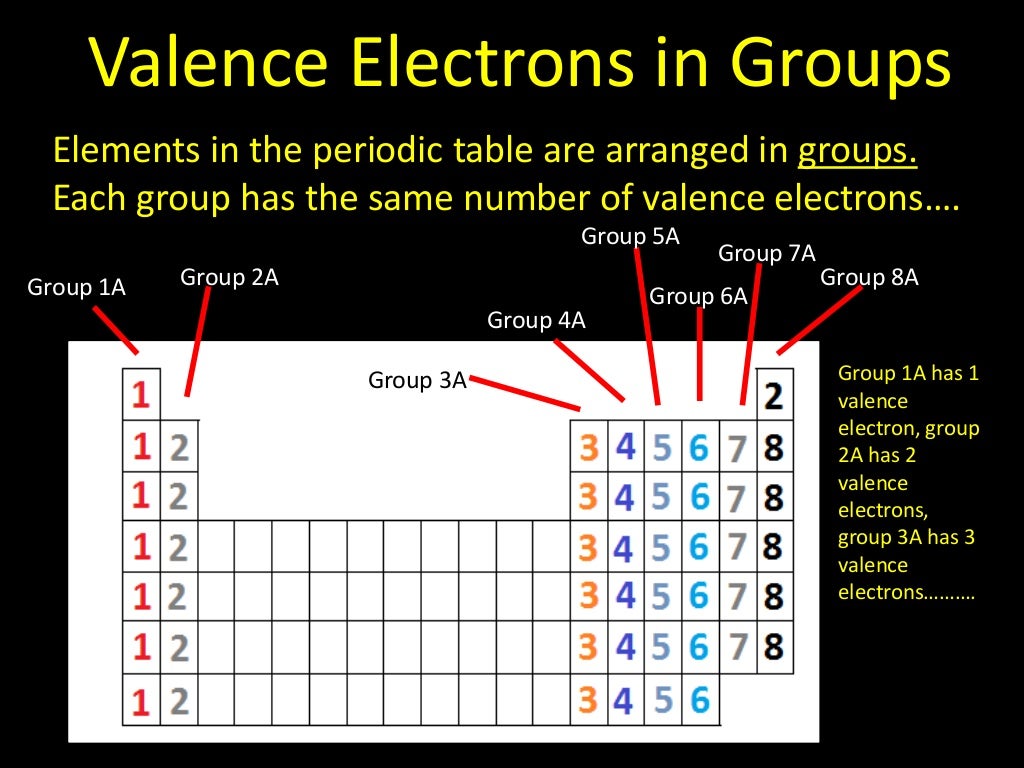
Please note that the table above provides a concise overview of the number of valence electrons for a few selected elements. Now, let’s take a look at some key takeaways about valence electrons: Key Takeaways Element Number of Valence Electrons Hydrogen 1 Oxygen 6 Carbon 4 Sodium 1 Chlorine 7 Understanding the number and arrangement of valence electrons is crucial in predicting the behavior of elements and their compounds. The valence electron determines the atom‘s reactivity and its ability to form chemical bonds. It refers to the outermost electron of an atom, which is involved in the atom‘s chemical reactions and bonding with other atoms. The valence electron is an important concept in chemistry and physics. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
